Darzalex is a drug that contains daratumumab, an antibody that binds to a protein known as CD38, which is present in multiple myeloma cells, causing the immune system to attack cells more effectively.
This medicine can not be bought in conventional pharmacies because it should only be given in the hospital by a doctor or nurse.

Price
Darzalex can not yet be purchased in Brazil, as it does not have Anvisa approval. However, studies are being done to get the drug approved.
What is it for
It is indicated for the treatment of adults with relapsed or relapsed multiple myeloma for whom treatment with a protease inhibitor and an immunomodulator has already been done and has shown pre-selection of the disease to the previous treatment.
How to use
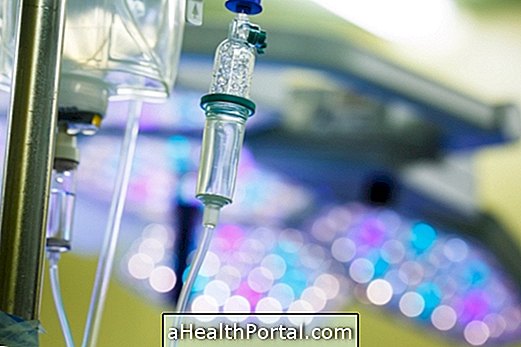
Darzalex should only be used in the hospital and administered by a healthcare professional as it is necessary to use other medicines before and after administration to reduce the risk of side effects.
The general recommendations indicate a dose of 16 mg / kg of body weight, being administered by infusion directly into the vein and following the following scheme:
- Weeks 1 to 8 : 1 time per week;
- Weeks 9 to 24 : 1 time every 2 weeks;
- From week 25 : 1 time per month.
Treatment with this medicine should be discontinued after week 25 if signs of developing the disease appear.
Possible side effects
Some of the most common side effects include respiratory infections, anemia, changes in blood test, fever, shivering headaches, decreased appetite, increased blood pressure, cough, stuffy nose, feeling of shortness of breath, nausea, diarrhea, arrest constipation, vomiting, joint pain and excessive tiredness.
Who should not use
Darzalex should not be used in pregnant or nursing women, children or anyone with an allergy to any of the ingredients in the formula. In addition, it should be used with caution in patients with liver and kidney problems.